The most pressing biological challenges to eradication include the development of drug and insecticide resistance, insufficiently sensitive parasite detection methods, limited effectiveness of standard vector control interventions in areas with intense malaria transmission and where outdoor biting is common, and the spillover of zoonotic simian malaria into humans. Fortunately, tools with strong potential to overcome these challenges are either already in the malaria community’s arsenal or are rolling out, and the research and development pipeline for new technologies has never been stronger.
Molecular methods for diagnosis and surveillance, novel drugs and insecticides, and a malaria vaccine have all become available in recent years. The research and development pipeline is expected to yield additional new drugs and insecticides, innovative vector control strategies, and more sensitive and precise diagnostics over the coming decade. In the longer term, gene drive technologies have potential to radically reduce transmission in the most challenging settings.
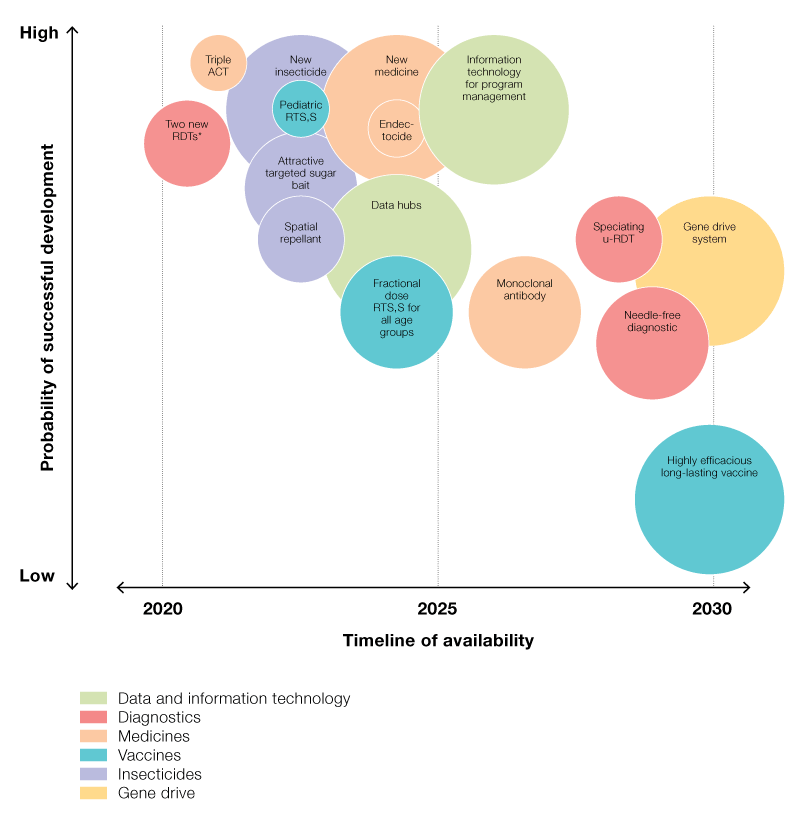
Research and development targets for malaria eradication that are most promising and impactful are shown in Figure 6. New tools will be particularly valuable if they improve surveillance, have long durations of efficacy, do not require difficult or protracted compliance from individuals, counter-drug and insecticide resistance, and are able to reduce malaria in high transmission settings.
Figure 6 ([in full report]) - Research and development framework for malaria eradication